 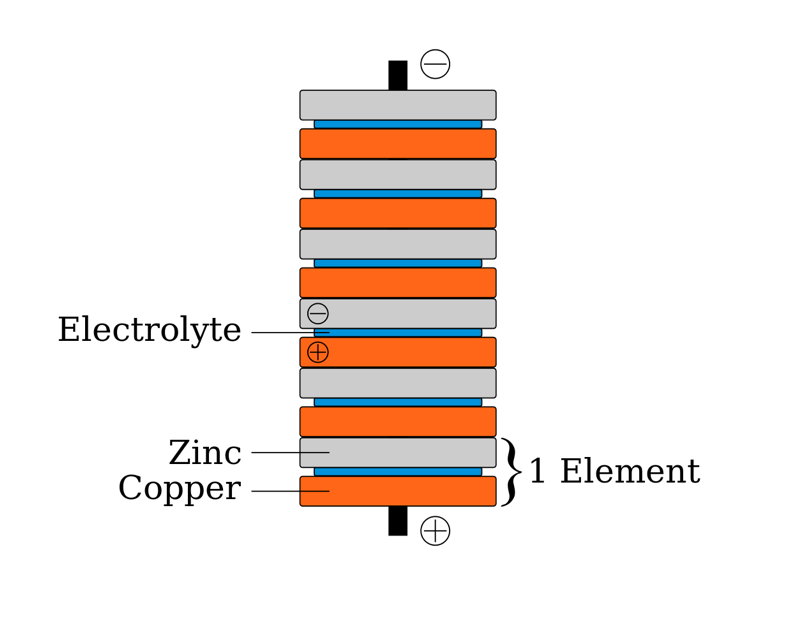
If the energy is the amount of water and your river is narrow, it needs to flow with higher speed or current to have the same energy/water supplied as if it’s a wide river but the current is flowing slowly. To make use of it for an electronic device, you need to choose a medium that is doing a good job at holding a lot of electrons and at making them flow to the other side, which creates what is called a current, like a river current that flows steadily in one direction. What sounds like names out of a Greek drama is as simple as that. Because having a few hundred electric eels in a tank in your car to power it is not a working plan, a different medium was searched for to hold electrons, and that was metal.Īn anode and cathode are nothing other than a place where we have more or fewer electrons in a medium. The famous inventor Nikola Tesla wanted to make nature a part of the electricity supply, and although he proved it works, he had too many enemies and no commercial model to scale it. Nature is diverse and some animals make use of electricity like for instance the electric eel but there are many more. The reason why we call a battery a battery is simply because we designed them to be able to store more electrons than we usually find in nature to make use of it. Whoever has had their hair stand up or got an electric hit from a cow fence or another person knows how it feels when electrons suddenly flow – it can hurt.īatteries surround us and we are sometimes batteries too, therefore, batteries are everywhere and normal. Even we humans sometimes behave like a battery. The anode and cathode are just the two names that describe the place where the electrons either move to or come from.Īll and everything in our world has electrons, and all and everything has at a certain point in time more electrons or fewer electrons, behaving like a battery. Forget all sophisticated technical terminology and measures, because that is all you need to know. You either make energy available for whatever electric device you use it for, or you put energy in the medium to store it for later use. While you do that, you charge or discharge a battery with energy. Our immediate next steps will be the improvement of the hydroflux process based on our understanding of the reaction pathway.What the heck is an anode and cathode in a battery, and why is that important?Ī battery is nothing other than a medium in which you move electrons from one side to the other. "Our development of this hydroflux process will enable energy saving measures in various ceramic production processes. 
"This work is the first experimental demonstration of the thermochemical stability of layered LiCoO 2 at low temperatures under ambient pressure," concludes Matsui. The team also measured the electrochemical properties of the layered LiCoO 2, showing that they were only marginally inferior to that of commercially available LiCoO 2 synthesized by the traditional high temperature method. "Specifically, the presence of water molecules in the starting materials significantly improved crystallinity of the end product." "By understanding the reaction pathway, we were able to identify the factors that promoted the crystal growth of layered LiCoO 2," Matsui said. The process was called the "hydroflux process." They were also able to determine the reaction pathway that led to the formation of the layered crystals. Using cobalt hydroxide and lithium hydroxide as starting materials, with sodium or potassium hydroxide as an additive, the team conducted a series of high-precision experiments under varying conditions to synthesize layered LiCoO 2 crystals. The layered LiCoO 2 is used in Li-ion batteries." "One form is layered rocksalt structure, called the high-temperature phase, and the other form is spinel-framework structure, called the low-temperature phase. "Lithium cobalt oxide can typically be synthesized in two forms," Matsui explains. Their findings were published in the journal Inorganic Chemistry. Traditionally, the synthesis of this compound requires temperatures over 800☌ and takes 10 to 20 hours to complete.Ī team of researchers at Hokkaido University and Kobe University, led by Professor Masaki Matsui at Hokkaido University's Faculty of Science, have developed a new method to synthesize lithium cobalt oxide at temperatures as low as 300☌ and durations as short as 30 minutes. Lithium cobalt oxide (LiCoO 2) is the compound used for the cathode in LIB for handheld electronics. Lithium ion batteries (LIB) are the most commonly used type of battery in consumer electronics and electric vehicles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
